Global Dietary Supplements Testing Market Size
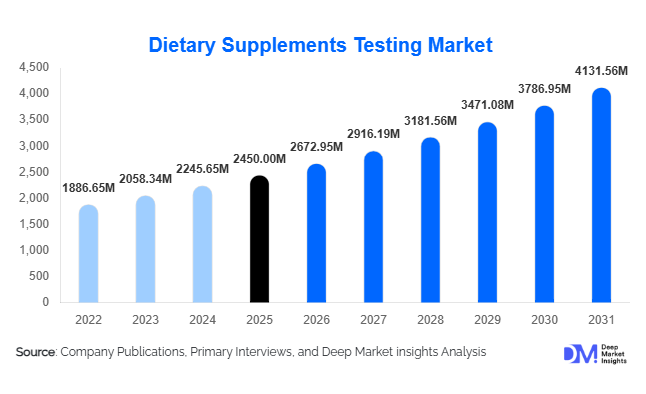
According to Deep Market Insights,the global dietary supplements testing market size was valued at USD 2,672.95 million in 2026 and is projected to grow from USD 2,450 million in 2026 to reach USD 4,131.56 million by 2031, expanding at a CAGR of 9.1% during the forecast period (2026–2031). The market growth is primarily driven by rising global consumption of dietary supplements, stringent regulatory requirements across North America, Europe, and Asia-Pacific, and increasing demand for advanced testing services to ensure product safety, quality, and authenticity.
Key Market Insights
- Regulatory compliance and quality assurance are the core drivers, with manufacturers relying on independent third-party laboratories for testing contaminants, heavy metals, microbiological safety, and ingredient potency.
- Technological integration, including chromatography, mass spectrometry, and molecular diagnostics, is enhancing accuracy, turnaround time, and traceability, making testing a strategic part of product validation.
- Asia-Pacific is the fastest-growing region, with China and India emerging as manufacturing and export hubs for supplements, driving demand for globally accredited testing services.
- North America leads the market, driven by high supplement consumption, strict FDA regulations, and increasing consumer awareness of product safety.
- Europe shows strong demand growth, particularly for herbal, botanical, and clean-label products that require rigorous testing for authenticity and contamination.
- Export-oriented manufacturing and cross-border trade are boosting testing volumes globally, as brands comply with multi-region regulatory standards to access international markets.
What are the latest trends in the dietary supplements testing market?
Advanced Analytical Technology Adoption
Laboratories are increasingly deploying high-resolution chromatography, mass spectrometry, and PCR-based methods to verify ingredient potency, detect contaminants, and authenticate botanical products. Automation and AI-assisted spectral analysis are improving accuracy and efficiency, reducing turnaround time for manufacturers. Rapid testing kits and digital reporting platforms are also being implemented to provide real-time results and traceability, enhancing transparency for both regulators and consumers. These innovations are particularly critical for high-demand segments such as probiotics, herbal supplements, and plant-based nutraceuticals.
Third-Party Certification and Brand Trust
Consumer demand for transparency and clean-label products has heightened the need for third-party testing and certification. Brands increasingly market “lab-verified” or “tested for purity” claims, making independent laboratories essential partners. This trend is particularly strong among e-commerce and export-driven brands, where credibility impacts purchase decisions. Accreditation from ISO, NSF, and other recognized bodies further strengthens trust and market competitiveness.
What are the key drivers in the dietary supplements testing market?
Rising Global Supplement Consumption
Expanding awareness of preventive healthcare and nutrition is driving higher consumption of vitamins, minerals, herbal supplements, protein powders, and probiotics. This growth increases demand for testing services to ensure safety, regulatory compliance, and quality assurance for both domestic and export markets. The rapid adoption of supplements in emerging economies, along with e-commerce growth, is particularly fueling market expansion.
Stringent Regulatory Oversight
Authorities worldwide, including the FDA (U.S.), EFSA (Europe), and FSSAI (India), have intensified regulations on contaminants, heavy metals, labeling accuracy, and microbial safety. Mandatory testing and batch certification have become prerequisites for market entry, compelling manufacturers to rely on sophisticated laboratory testing. Regulatory changes often drive recurring testing demand, creating long-term market growth.
Concern over Product Adulteration
Botanical, herbal, and protein supplements are prone to adulteration and substitution. High-profile product recalls and contamination cases have made testing critical for brand protection. Laboratories offering contaminant detection, DNA authentication, and potency validation are seeing increased demand, positioning testing services as essential for risk mitigation.
What are the restraints for the global market?
High Capital Expenditure Requirements
Setting up advanced laboratories with chromatography, mass spectrometry, and molecular diagnostic platforms requires substantial investment. High operational costs, skilled workforce requirements, and maintenance expenses can deter smaller players from entering the market and limit expansion opportunities for some laboratories.
Competition from In-House Testing Facilities
Large supplement manufacturers are increasingly establishing in-house testing labs to reduce costs and control quality. This vertical integration can limit third-party laboratory revenues, particularly in mature markets such as North America and Europe. Balancing pricing and service differentiation remains a challenge for independent providers.
What are the key opportunities in the dietary supplements testing market?
Export Compliance and Multi-Region Testing
Manufacturers exporting to the U.S., Europe, and Asia require multi-standard certifications. Laboratories offering cross-regulatory testing services for heavy metals, contaminants, and potency can secure long-term contracts with global brands. Emerging economies in Asia-Pacific and Latin America are increasingly exporting supplements, creating high-value testing opportunities.
Specialty Product Validation
The growing demand for plant-based, vegan, and organic supplements drives the need for botanical verification, DNA barcoding, and isotope ratio testing. As consumers seek transparency and authenticity, laboratories offering these high-complexity services can differentiate themselves and capture premium revenue.
Integration of Advanced Analytical Technologies
AI-assisted spectral analysis, automation, and rapid pathogen detection platforms present opportunities to improve turnaround time and accuracy. Laboratories investing in these technologies can cater to high-volume clients efficiently and reduce operational costs, boosting profitability and market share.
Product Type Insights
Herbal & botanical supplements dominate the dietary supplements testing market, accounting for nearly 24% of global testing volumes in 2025. The leadership of this segment is primarily driven by the high risk of adulteration, substitution, and contamination associated with plant-based raw materials, which necessitates frequent authenticity verification, heavy metal screening, pesticide residue analysis, and DNA-based identification. Increasing global consumption of botanical extracts, Ayurvedic ingredients, and traditional medicine formulations has intensified regulatory oversight, thereby accelerating testing frequency and depth. Vitamins & minerals testing follows closely, supported by mandatory batch-wise chemical composition analysis, potency validation, and stability verification to ensure label claim compliance. Protein supplements, probiotics, and multifunctional blends are witnessing robust growth due to rising sports nutrition demand, gut health awareness, and personalized nutrition trends. These complex formulations require microbiological validation, strain identification, amino acid profiling, and allergen testing, prompting laboratories to expand their analytical capabilities through advanced chromatography, spectroscopy, and molecular diagnostic technologies. As product innovation accelerates globally, laboratories are diversifying service portfolios to support multi-ingredient formulations and international compliance standards.
Application Insights
Nutritional quality verification remains the largest application segment, driven by stringent regulatory mandates requiring accurate potency, nutrient content validation, and label claim substantiation. With rising consumer scrutiny and transparency expectations, brands increasingly rely on third-party laboratories to validate ingredient concentrations and ensure batch-to-batch consistency. Contaminant testing represents another major application area, as global regulators enforce strict limits on heavy metals, pesticide residues, microbial contamination, and solvent residues. Authentication testing is expanding rapidly, particularly for botanical, organic, vegan, and non-GMO claims, where DNA barcoding and advanced fingerprinting techniques are employed to prevent fraud and protect brand reputation. Stability and shelf-life analysis further support formulation optimization, packaging validation, and regulatory compliance across different climatic zones. Additionally, rapid testing solutions and digital reporting platforms are gaining traction among export-oriented and e-commerce-driven brands that require quick turnaround times, traceability documentation, and cost-efficient compliance management systems.
Distribution Channel Insights
Direct B2B contracts with supplement manufacturers account for the majority of market revenues, as laboratories provide recurring batch testing, certification, audit support, and export compliance documentation. The dominance of this channel is driven by long-term contractual relationships and the need for continuous quality monitoring throughout production cycles. Contract research organizations are increasingly offering outsourced analytical services to nutraceutical brands and private-label producers seeking flexible and scalable testing infrastructure. Digital transformation is reshaping distribution channels, with laboratories implementing online portals and Laboratory Information Management Systems (LIMS) that enable remote sample submission, real-time tracking, and automated reporting. Export-focused testing packages that bundle multi-country regulatory certifications are emerging as premium service offerings, particularly for companies targeting highly regulated markets such as North America and Europe.
End-Use Insights
Nutraceutical manufacturers represent over 55% of total testing demand globally, driven by expanding domestic consumption, export compliance requirements, and growing regulatory oversight. The leading position of this segment is supported by high production volumes and the necessity for routine quality validation across diverse product portfolios. Contract manufacturers are the fastest-growing end-use segment, benefiting from the rapid expansion of private-label brands and cross-border e-commerce channels that require certified third-party validation. Pharmaceutical companies producing over-the-counter supplements are adopting pharmaceutical-grade testing standards, further increasing demand for high-precision analytical services. Retail and e-commerce brands increasingly leverage independent laboratory certifications to strengthen consumer trust and differentiate products in competitive markets. Asia-Pacific manufacturing hubs, particularly China and India, are expanding export volumes across sports nutrition, probiotics, and functional foods categories, thereby increasing multi-standard testing requirements across end-use industries.
| By Product Type Tested | By Testing Type | By Service Provider Type | By Technology & Analytical Method | By End-Use Industry |
|---|---|---|---|---|
|
|
|
|
|
Regional Insights
North America
North America holds around 34% of the global dietary supplements testing market share in 2025, with the United States contributing approximately 28%. Market leadership is driven by strong regulatory enforcement under the U.S. Food and Drug Administration framework, high per-capita dietary supplement consumption, and growing consumer preference for certified and third-party-verified products. Increasing litigation risks and product recall awareness further encourage manufacturers to invest in comprehensive testing programs. Canada also represents a significant contributor, where compliance with Health Canada regulations and Natural Health Product standards requires rigorous testing protocols. The region benefits from advanced laboratory infrastructure, widespread adoption of digital reporting systems, and a mature nutraceutical industry that prioritizes transparency and traceability.
Europe
Europe accounts for approximately 27% of the global market in 2025, led by Germany, the United Kingdom, France, and Italy. Regional growth is supported by strict European Food Safety Authority guidelines, harmonized regulatory frameworks, and increasing demand for clean-label, organic, and sustainably sourced supplements. Botanical and plant-based products require extensive authenticity and contaminant testing, particularly due to cross-border trade within the European Union. Younger consumers’ preference for certified and ethically sourced supplements, coupled with rising vegan and plant-based lifestyles, further drives laboratory testing demand. High market maturity, recurring compliance contracts, and established quality standards sustain steady regional growth.
Asia-Pacific
Asia-Pacific is the fastest-growing regional market, fueled by expanding manufacturing activities in China and India, which serve as major global export hubs for nutraceutical ingredients and finished supplements. Rapid industrialization, government-backed manufacturing initiatives, and increasing export scrutiny from Western markets are intensifying demand for multi-parameter testing services. Japan and South Korea demonstrate stable domestic consumption growth, particularly in premium supplements and functional foods, requiring advanced quality verification processes. The rapid expansion of e-commerce platforms and cross-border online supplement sales is accelerating the adoption of rapid testing, digital certification, and internationally recognized compliance standards across the region.
Latin America
Latin America contributes approximately 8% of the global market, with Brazil and Mexico emerging as key growth centers. Regional expansion is supported by rising health awareness, increasing disposable income, and growing adoption of preventive healthcare supplements. Regulatory frameworks are gradually strengthening, encouraging manufacturers to adopt standardized testing procedures. Export-oriented producers are increasingly investing in third-party certifications to access North American and European markets. While the market remains relatively developing compared to mature regions, improving regulatory enforcement and expanding domestic supplement production are steadily driving testing demand.
Middle East & Africa
The Middle East & Africa region accounts for approximately 6–7% of the global market, with the United Arab Emirates and South Africa serving as primary contributors. Growth is driven by increasing consumer awareness regarding product safety, rising imports of dietary supplements, and expanding retail and e-commerce channels. Regulatory authorities in the Gulf Cooperation Council countries are strengthening quality standards, prompting importers and distributors to seek certified testing services. In Africa, improving healthcare infrastructure and gradual regulatory formalization are encouraging laboratory investments and third-party verification practices. Intra-regional trade and growing wellness trends continue to create long-term growth opportunities for testing providers.
| North America | Europe | APAC | Middle East and Africa | LATAM |
|---|---|---|---|---|
|
|
|
|
|
Key Players in the Dietary Supplements Testing Market
- Eurofins Scientific
- SGS SA
- Bureau Veritas
- Intertek Group plc
- TÜV SÜD
- ALS Limited
- NSF International
- UL Solutions
- Mérieux NutriSciences
- AsureQuality
- Romer Labs
- Microbac Laboratories
- Campden BRI
- Covance Inc.
- Tentamus Group